|
This video also goes through some notes on reactivity trends. This video will go through patterns of reactivity for group 1. Hence the electronegativity and the ionisation energy determine a chemical reaction. 
Reactivity generally increases from the centre of the periodic table to the left (recall the right-hand side has non-metals), and also from the top of the periodic table down. The higher the reactivity of the element, the easier it is to combine. See if you can apply your knowledge of the ionisation energy (energy required to expel an electron from an atom), electronegativity (ability to attract electrons to atom) and atomic radii (half the distance between two neighbouring nuclei of the same element) trends on the periodic table. This video will go through some general reactivity trends across the periodic table. 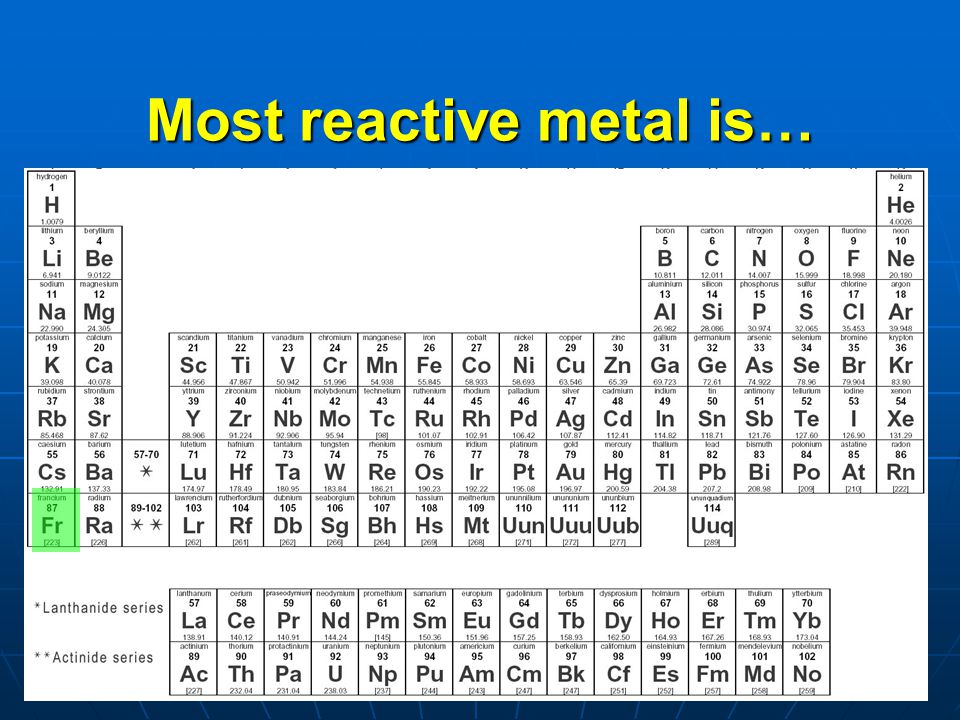
Analyse patterns in metal activity on the periodic table and explain why they correlate with, for example:.This post will go through some patterns in metal activity for Prelim Chemistry. 658 Likes, TikTok video from Your Tutor (yourtutor12): 'Reactivity of group 1 -the alkali metals group1 reactivity periodictable learnontiktok. Metals and non-metals have different properties and different reactions. What Are Some Patterns in Metal Activity?
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
